Teva Branded Pharmaceutical Products R&D;, . An electronic inhaler add-on module which store and transmit information related to inhaler use (eModule) PDR0000401 FCC ID 2AJVSPDR0000401
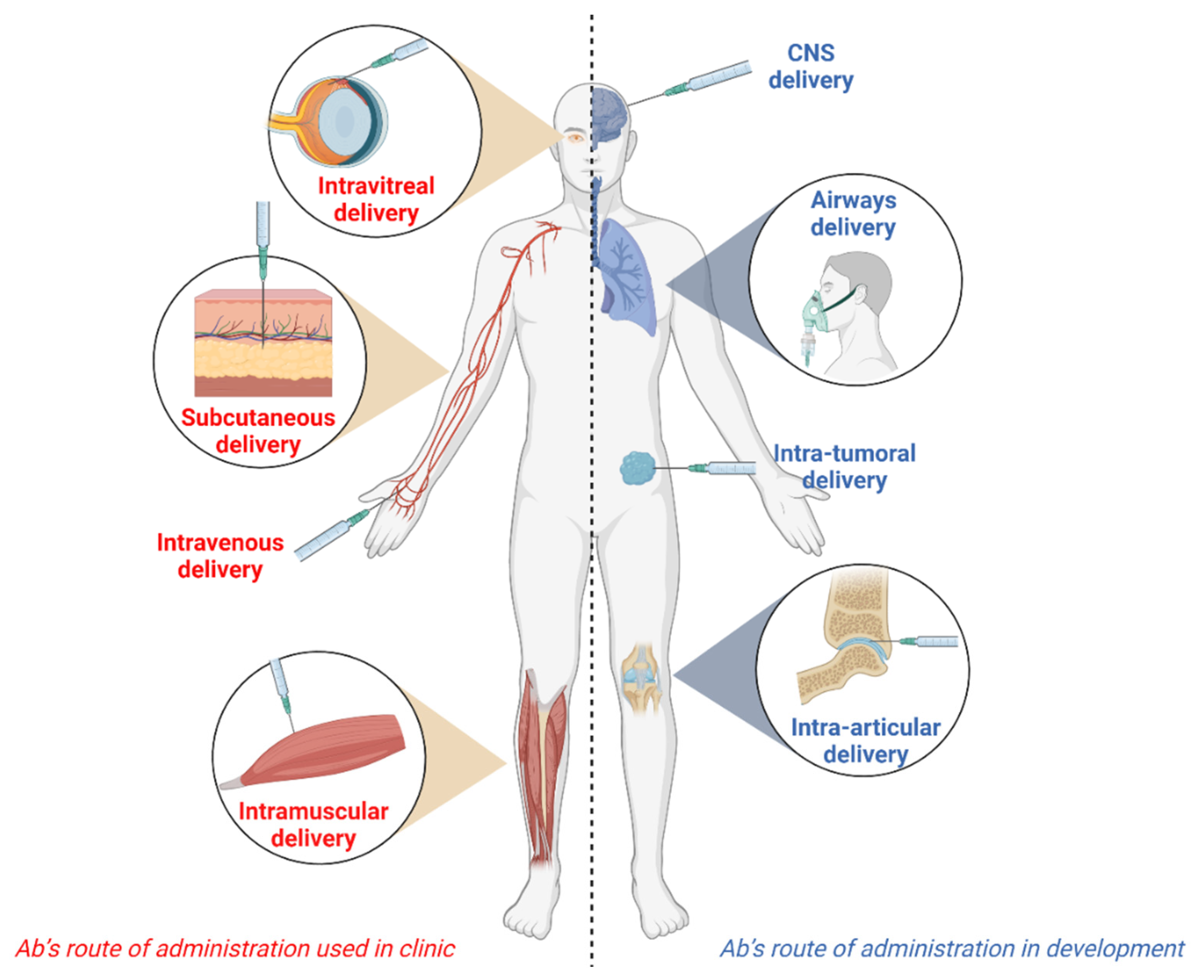
Antibodies | Free Full-Text | Alternative Routes of Administration for Therapeutic Antibodies—State of the Art
1 UNITED STATES DISTRICT COURT DISTRICT OF NEW JERSY IN RE: ELMIRON (PENTOSAN POLYSULFATE SODIUM) PRODUCTS LIABILITY LITIGATION
Investigating the Accuracy of the Digihaler, a New Electronic Multidose Dry-Powder Inhaler, in Measuring Inhalation Parameters
NAPOLI SHKOLNIK PLLC BY: HUNTER J. SHKOLNIK, ESQUIRE N.J. BAR NO: 41531985 400 Broadhollow Rd. Suite 305 Melville, NY 11747 Phon

Natalia Wojcik - Associate Director, Global New Product Introduction - Teva Pharmaceuticals | LinkedIn

Teva Branded Pharmaceutical Products R&D;, . An electronic inhaler add-on module which store and transmit information related to inhaler use (eModule) PDR0000401 FCC ID 2AJVSPDR0000401
A Comparison of the Pharmacokinetic Profile of an Ascending-Dose, Extended-Regimen Combined Oral Contraceptive to Those of Other
Predicting Responders to Reslizumab after 16 Weeks of Treatment Using an Algorithm Derived from Clinical Studies of Patients wit