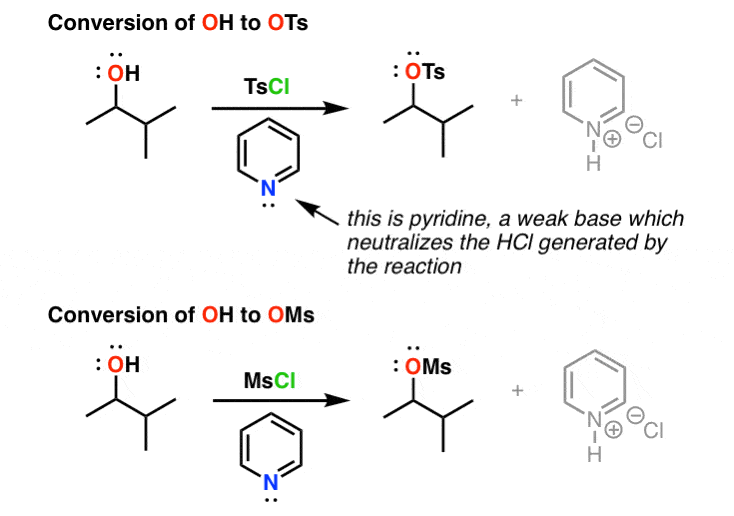
Reagent Friday: TsCl (p-toluenesulfonyl chloride) and MsCl (methanesulfonyl chloride) – Master Organic Chemistry

Reaction scheme. (i) p -toluenesulfonyl chloride, TEA, MDC, r. t., 4 hr. | Download Scientific Diagram

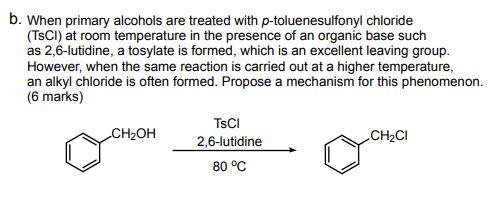
What is the mechanism for the following alcohol with p-TsCl/pyridine followed by addition of a strong base | Homework.Study.com

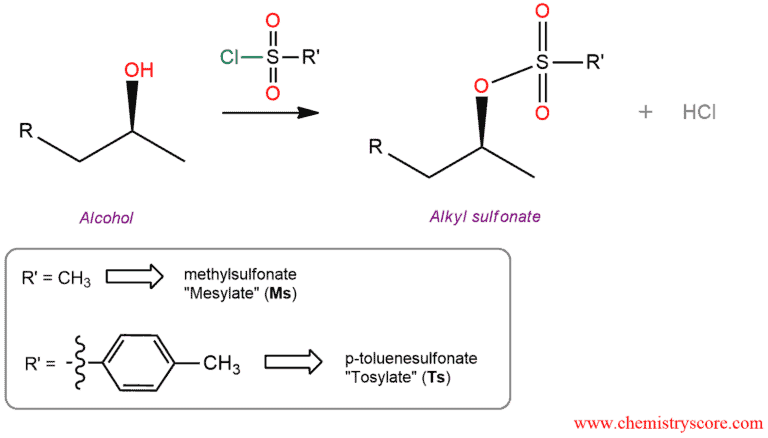
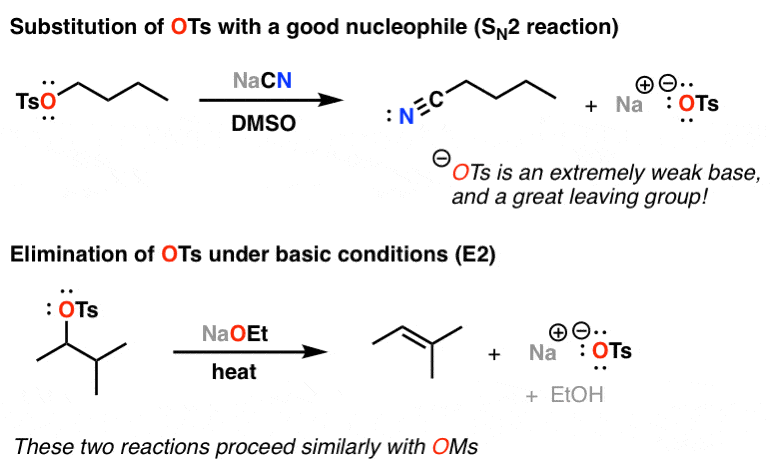
Alcohols are converted to tosylates by treatment with p-toluence sulfonyl chloride (TsCl) in the presence of pyridine. This overall process converts a poor leaving group (overset(Ө)H) into good one (overset(Ө)Ts). A tosylate

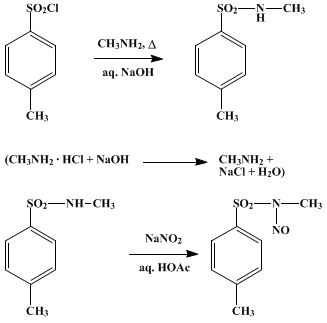
N-Sulfonylation of amines, imides, amides and anilides using p-TsCl in presence of atomized sodium in EtOH–THF under sonic condition - ScienceDirect
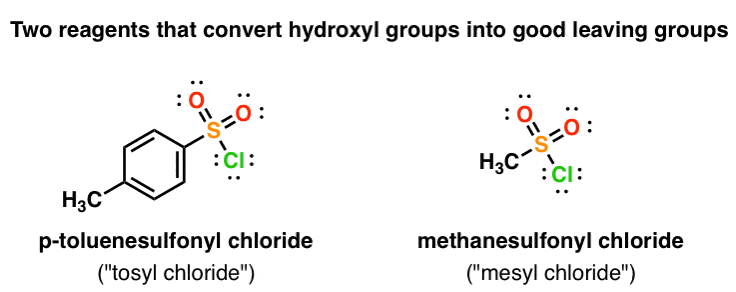
Reagent Friday: TsCl (p-toluenesulfonyl chloride) and MsCl (methanesulfonyl chloride) – Master Organic Chemistry

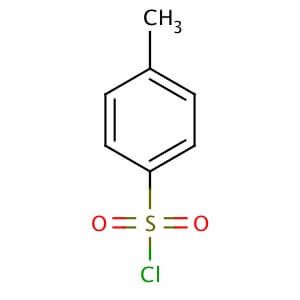
P-TOLUENESULFONYL CHLORIDE Formula - C7H7ClO2S - Over 100 million chemical compounds | Mol-Instincts