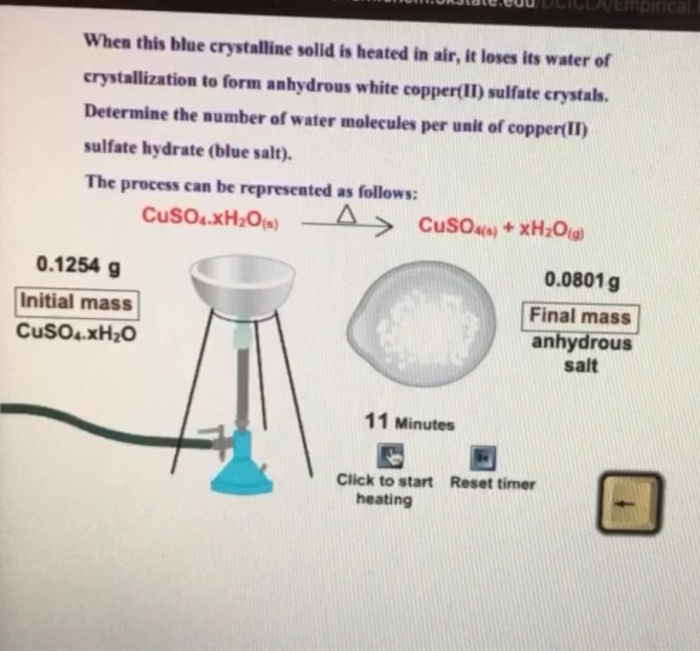
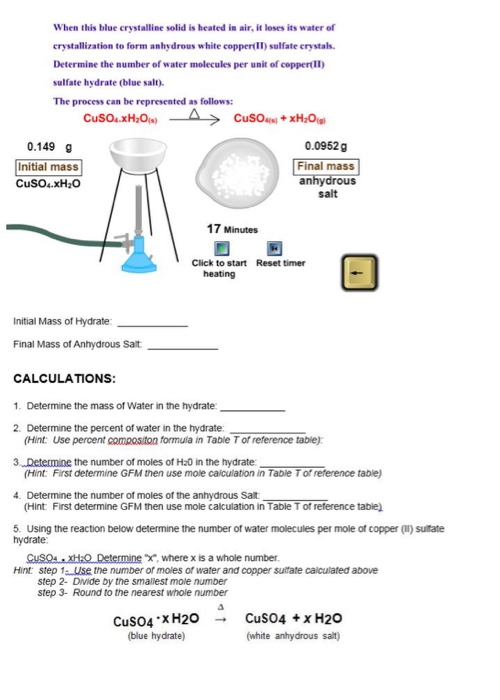
On strongly heating, a blue salt leaves a black residue. Which of the following cations can be present in the salt?

A blue - coloured salt becomes white on heating. Give reason for the above observation. What happens when we add water to the salt obtained after heating ? Also write its formula.

Homogeneous Metal Salt Solutions for Biomass Upgrading and Other Select Organic Reactions | ACS Catalysis

A blue - coloured salt becomes white on heating. Give reason for the above observation. What happens when we add water to the salt obtained after heating ? Also write its formula.

A blue - coloured salt becomes white on heating. Give reason for the above observation. What happens when we add water to the salt obtained after heating ? Also write its formula.
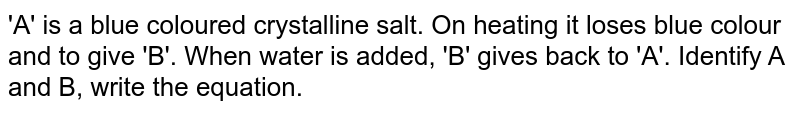
A blue - coloured salt becomes white on heating. Give reason for the above observation. What happens when we add water to the salt obtained after heating ? Also write its formula.

The metal salt A is blue in colour. When salt A is heated strongly over a burner, then a substance B - Brainly.in

Air-/Heat-Stable Crystalline Carbon-Centered Radicals Derived from an Annelated N-Heterocyclic Carbene | Journal of the American Chemical Society